Methanol Chemical
Product Details:
Methanol Chemical Price And Quantity
- 1 Ton
- 35 INR/Ton
Methanol Chemical Trade Information
- 50 Ton Per Day
- 2 Days
- All India
Product Description
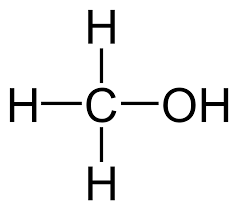
Methanol is a colourless, flammable liquid with the chemical formula CH3OH. Due to its historical creation via wood distillation, it is also known as wood alcohol and is the most basic type of alcohol. It has many uses, including as a fuel, antifreeze, solvent, and raw material for chemical synthesis. Methanol is necessary for the manufacturing of acetic acid and formaldehyde, two chemicals used in many different industries. Although it has certain industrial use, it is poisonous and can have a negative impact on one's health if consumed or inhaled. Due to its possible risks, methanol is utilised properly, ensuring handling is safe.
Methanol, which has the molecular formula CH3OH, has a number of qualities and advantages that make it useful in a variety of applications.
Features of Methanol Chemical:
1. Methanol has excellent solvent characteristics that enable it to dissolve a variety of polar and non-polar substances, facilitating chemical reactions and industrial uses.
2. High purity: It can be easily distilled to remove impurities, making it ideal for use in electronics and pharmaceuticals.
3. Low freezing point: Due to its low freezing point (-97.6 Deg C or -143.7 Deg F), it is a powerful antifreeze agent, notably in cooling systems and windscreen washer fluids.
4. Clean burning: Methanol burns cleanly and emits less hazardous gases than traditional fossil fuels, making it a more environmentally friendly fuel option.
Benefits of Methanol Chemical:
1. Chemical synthesis: The manufacturing of formaldehyde, acetic acid, and other chemicals requires the use of methanol as a key raw material. These compounds are used in a variety of industries, including those that produce plastics, textiles, and medicines.
2. Renewable energy: Methanol is a sustainable fuel alternative that may be made from a variety of renewable sources, including carbon dioxide and biomass.
3. Transportation fuel: Using methanol as a substitute fuel in cars helps cut greenhouse gas emissions, aiding in the fight against climate change.
4. Cost-effective: Methanol provides economical solutions for many industrial processes and applications because it is a widely accessible and reasonably priced chemical.
To avoid potential risks to human health and the environment, it is crucial to remember that methanol is poisonous and combustible, necessitating careful handling and the appropriate safety measures.
 |
ASHISH IMPEX
All Rights Reserved.(Terms of Use) Developed and Managed by Infocom Network Private Limited. |
 English
English Spanish
Spanish French
French German
German Italian
Italian Chinese (Simplified)
Chinese (Simplified) Japanese
Japanese Korean
Korean Arabic
Arabic Portuguese
Portuguese